Sohum innovation lab- realising the goal of “Accessible India”

While working in the child development clinic at All India Institute of Medical Sciences ( AIIMS), I came across a five year old child with an undetected hearing disability. Although universal screening is mandatory at the time of birth, in a resource-poor country like India, which has extensive demand-supply gaps in health care, there are cases when hearing impairment goes undiagnosed. If this impairment is not detected at an early stage, it can impede speech, language and cognition.Presently hearing screening is done with subjective tests such as questionnaires, observations or Oto-Acoustic-Emission (OAE), which have a high false-positive rate. 800,000 hearing impaired babies are born every year all over the world, of which 100,000 are in India and 90% are in developing countries. Besides India, 40 low income and 53 low middle income countries do not have an affordable solution for early screening of hearing impairment. We felt strongly about this situation and believed that it needed to be remedied. We set about on a six-month journey of rigorous field-research with physicians, designers and like-minded individuals to try and find a solution to this problem.
The outcome of our research was theSohum screening device, whichis based on the gold standard Brainstem Auditory Evoked Response (BAER or ABR technology), which measures auditory brain waves through electrodes placed on a baby’s head. The electrodes are stimulated to gauge electrical responses generated by the brain’s auditory system. If there is no response, the child cannot hear. This non-invasive device was made with an in-built algorithm that filters out ambient noises from the test signal.The optimized design reduces test duration by reducing the time required for preparation and analysis, making it ideal for mass screening. The electrode system is reusable, which reduces the cost of the procedure. The business model is a unique service based revenue sharing model.

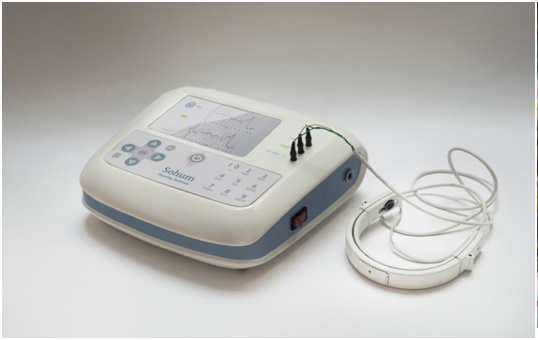
We have clearances for clinic trials from AIIMS and other clinical facilities to prove the efficacy of the device. A full PCT patent application has been filed. We have conducted clinical evaluation to prove the sensitivity and specificity of the device.
Currently, the Sohum device is installed in five hospitals – two in Bengaluru, one in Manipal, one in Delhi and one in Guatamelaand 175 successful tests have been conducted. Sohum Innovation has won many laurels; a testimony of the dire need for such devices in developing nations such as India.After due diligence by the country’s top scientists, the company was awarded funding support by the Biotechnology Industry Research Assistance Council (BIRAC)India in the form of the BIRAC Ignition Grant, Grand Challenges Canada, Center for Innovation in Global health, NASSCOM, Indian Merchant Chambers. Sohum provides early screening, that leads to timely treatment and rehabilitation, as well as savings in healthcare expenses to the system. We will be cross subsidizing the product and services to reach remote areas and non-institutional births.